Quantum Numbers
Quantum Numbers
Developed to better understand the movement and pathway of electrons in the designated orbital within an atom. It indicates an electrons trait within an atom, which satisfies to explain the movement of electron’s as a wave function.
There are four quantum numbers:
1.) The principal quantum number (n)
2.) The orbital angular momentum quantum number (l)
3.) The magnetic quantum number (ml)
4.) The electron spin quantum number(ms)
· Principal Quantum Number (n) – describes the energy of an electron and the probable distance of electron in the nucleus.
· Orbital Angular momentum (l) – describes the shape of the orbital.
· Magnetic Quantum Number (ml) – describes the amount of energy levels in a subshell.
· Electron Spin Quantum number (ms) – is referring to the spin on the electron, which either be up or down.
-Diana Rose R. Chico-
Developed to better understand the movement and pathway of electrons in the designated orbital within an atom. It indicates an electrons trait within an atom, which satisfies to explain the movement of electron’s as a wave function.
There are four quantum numbers:
1.) The principal quantum number (n)
2.) The orbital angular momentum quantum number (l)
3.) The magnetic quantum number (ml)
4.) The electron spin quantum number(ms)
· Principal Quantum Number (n) – describes the energy of an electron and the probable distance of electron in the nucleus.
· Orbital Angular momentum (l) – describes the shape of the orbital.
· Magnetic Quantum Number (ml) – describes the amount of energy levels in a subshell.
· Electron Spin Quantum number (ms) – is referring to the spin on the electron, which either be up or down.
-Diana Rose R. Chico-
Stoichiometry
Limiting and Excess Reagent
Limiting Reactant – The reactant in a chemical reaction that limits the amount of product that can be formed. The reaction will stop when all of the limiting reactant is consumed.
Excess Reactant - The reactant in a chemical reaction that remains when a reaction stops when the limiting reactant is completely consumed. The excess reactant remains because there is nothing with which it can react.
Sample Problem:
Sodium hydroxide (NaOH) reacts with phosphoric acid (H3PO4) to form sodium phosphate (Na3PO4) and water (H2O) by the reaction:
3 NaOH(aq) + H3PO4(aq) → Na3PO4(aq) + 3 H2O(l)
If 35.60 grams of NaOH is reacted with 30.80 grams of H3PO4,
a. How many grams of Na3PO4 are formed? b. What is the limiting reactant?
c. How many grams of the excess reactant remains when the reaction is complete?
Molar mass of NaOH = 40.00 grams
Molar mass of H3PO4 = 98.00 grams
Molar mass of Na3PO4 = 163.94 grams
Solution:
To determine the limiting reactant, calculate the amount of product formed by each reactant. The reactant the produces the least amount of product is the limiting reactant.
To determine the number of grams of Na3PO4 formed:
grams Na3PO4 = (grams reactant) x (mole of reactant/molar mass of reactant) x (mole ratio: product/reactant) x (molar mass of product/mole product)
Amount of Na3PO4 formed from 35.60 grams of NaOH
grams Na3PO4 = (35.60 g NaOH) x (1 mol NaOH/40.00 g NaOH) x (1 mol Na3PO4/3 mol NaOH) x (163.94 g Na3PO4/1 mol Na3PO4)
grams of Na3PO4 = 48.64 grams
Amount of Na3PO4 formed from 30.80 grams of H3PO4
grams Na3PO4 = (30.80 g H3PO4) x (1 mol H3PO4/98.00 grams H3PO4) x (1 mol Na3PO4/1 mol H3PO4) x (163.94 g Na3PO4/1 mol Na3PO4)
grams Na3PO4 = 51.52 grams
The sodium hydroxide formed less product than the phosphoric acid. This means the sodium hydroxide was the limiting reactant and 48.64 grams of sodium phosphate is formed.
To determine the amount of excess reactant remaining, the amount used is needed.
grams of reactant used = (grams of product formed) x (1 mol of product/molar mass of product) x (mole ratio of reactant/product) x (molar mass of reactant)
grams of H3PO4 used = (48.64 grams Na3PO4) x (1 mol Na3PO4/163.94 g Na3PO4) x (1 mol H3PO4/1 mol Na3PO4) x (98 g H3PO4/1 mol)
grams of H3PO4 used = 29.08 grams
This number can be used to determine the remaining amount of excess reactant.
Grams H3PO4 remaining = initial grams H3PO4 - grams H3PO4 used
grams H3PO4 remaining = 30.80 grams - 29.08 grams
grams H3PO4 remaining = 1.72 grams
Answer:
When 35.60 grams of NaOH is reacted with 30.80 grams of H3PO4,
a. 48.64 grams of Na3PO4 are formed.
b. NaOH was the limiting reactant.
c. 1.72 grams of H3PO4 remain at completion.
-Maria Simonne T. Oliveria
Limiting Reactant – The reactant in a chemical reaction that limits the amount of product that can be formed. The reaction will stop when all of the limiting reactant is consumed.
Excess Reactant - The reactant in a chemical reaction that remains when a reaction stops when the limiting reactant is completely consumed. The excess reactant remains because there is nothing with which it can react.
Sample Problem:
Sodium hydroxide (NaOH) reacts with phosphoric acid (H3PO4) to form sodium phosphate (Na3PO4) and water (H2O) by the reaction:
3 NaOH(aq) + H3PO4(aq) → Na3PO4(aq) + 3 H2O(l)
If 35.60 grams of NaOH is reacted with 30.80 grams of H3PO4,
a. How many grams of Na3PO4 are formed? b. What is the limiting reactant?
c. How many grams of the excess reactant remains when the reaction is complete?
Molar mass of NaOH = 40.00 grams
Molar mass of H3PO4 = 98.00 grams
Molar mass of Na3PO4 = 163.94 grams
Solution:
To determine the limiting reactant, calculate the amount of product formed by each reactant. The reactant the produces the least amount of product is the limiting reactant.
To determine the number of grams of Na3PO4 formed:
grams Na3PO4 = (grams reactant) x (mole of reactant/molar mass of reactant) x (mole ratio: product/reactant) x (molar mass of product/mole product)
Amount of Na3PO4 formed from 35.60 grams of NaOH
grams Na3PO4 = (35.60 g NaOH) x (1 mol NaOH/40.00 g NaOH) x (1 mol Na3PO4/3 mol NaOH) x (163.94 g Na3PO4/1 mol Na3PO4)
grams of Na3PO4 = 48.64 grams
Amount of Na3PO4 formed from 30.80 grams of H3PO4
grams Na3PO4 = (30.80 g H3PO4) x (1 mol H3PO4/98.00 grams H3PO4) x (1 mol Na3PO4/1 mol H3PO4) x (163.94 g Na3PO4/1 mol Na3PO4)
grams Na3PO4 = 51.52 grams
The sodium hydroxide formed less product than the phosphoric acid. This means the sodium hydroxide was the limiting reactant and 48.64 grams of sodium phosphate is formed.
To determine the amount of excess reactant remaining, the amount used is needed.
grams of reactant used = (grams of product formed) x (1 mol of product/molar mass of product) x (mole ratio of reactant/product) x (molar mass of reactant)
grams of H3PO4 used = (48.64 grams Na3PO4) x (1 mol Na3PO4/163.94 g Na3PO4) x (1 mol H3PO4/1 mol Na3PO4) x (98 g H3PO4/1 mol)
grams of H3PO4 used = 29.08 grams
This number can be used to determine the remaining amount of excess reactant.
Grams H3PO4 remaining = initial grams H3PO4 - grams H3PO4 used
grams H3PO4 remaining = 30.80 grams - 29.08 grams
grams H3PO4 remaining = 1.72 grams
Answer:
When 35.60 grams of NaOH is reacted with 30.80 grams of H3PO4,
a. 48.64 grams of Na3PO4 are formed.
b. NaOH was the limiting reactant.
c. 1.72 grams of H3PO4 remain at completion.
-Maria Simonne T. Oliveria
Molecular structure of the atom
Molecular structure of the atom
Molecular structure of the atom is a way of representing how elements are bonded using the electron dot formula or the Lewis structure
Lewis structure- uses dots to represent the electron being shared or transferred.
*Trivia about Gilbert N. Lewis
- did you know that Lewis studied here in the Philippines and also in Germany
There are two types of chemical bonding of electron one is ionic bonding is the transferring of electron from one element to another this type of chemical bond require a metal and a non-metal and its polarity is 2.0 and above the other one is covalent bonding is sharing of electrons both non-metals are needed.
Electronegativity-ability of the electron to attract electron other element
VSEPR - valence-shell electron-pair repulsion
*remember the most electronegative element is the central atom of a compound because of its ability to attract other element
Examples:
Formaldehyde is a toxic organic molecule with molecular formula CH2O. Draw the Lewis structure of formaldehyde.
Solution:
Step 1: Find the total number of valence electrons.
Carbon has 4 valence electrons
Hydrogen has 1 valence electrons
Oxygen has 6 valence electrons
Total valence electrons = 1 carbon (4) + 2 hydrogen (2 x 1) + 1 oxygen (6)
Total valence electrons = 12
Step 2: Find the number of electrons needed to make the atoms "happy"
Carbon needs 8 valence electrons
Hydrogen needs 2 valence electrons
Oxygen needs 8 valence electrons
Total valence electrons to be "happy" = 1 carbon (8) + 2 hydrogen (2 x 2) + 1 oxygen (8)
Total valence electrons to be "happy" = 20
Step 3: Determine the number of bonds in the molecule.
number of bonds = (Step 2 - Step 1)/2
number of bonds = (20 - 12)/2
number of bonds = 8/2
number of bonds = 4
Step 4: Choose a central atom.
Hydrogen is the least electronegative of the elements, but hydrogen is rarely the central atom in a molecule. The next lowest electronegative atom is carbon.
Step 5: Draw a skeletal structure.
Connect the other three atoms to the central carbon atoms. Since there are 4 bonds in the molecule, one of the three atoms will bond with a double bond. Oxygen is the only choice in this case, since hydrogen only has one electron to share.
Step 6: Place electrons around outside atoms.
There are 12 valence atoms total. Eight of these electrons are tied up in bonds. The remaining four complete the octet around the oxygen atom.
Each atom in the molecule has a complete outer shell full of electrons. There are no electrons left over and the structure is complete. The finished structure appears in the picture at the beginning of the example.
Molecular structure of the atom is a way of representing how elements are bonded using the electron dot formula or the Lewis structure
Lewis structure- uses dots to represent the electron being shared or transferred.
*Trivia about Gilbert N. Lewis
- did you know that Lewis studied here in the Philippines and also in Germany
There are two types of chemical bonding of electron one is ionic bonding is the transferring of electron from one element to another this type of chemical bond require a metal and a non-metal and its polarity is 2.0 and above the other one is covalent bonding is sharing of electrons both non-metals are needed.
Electronegativity-ability of the electron to attract electron other element
VSEPR - valence-shell electron-pair repulsion
*remember the most electronegative element is the central atom of a compound because of its ability to attract other element
Examples:
Formaldehyde is a toxic organic molecule with molecular formula CH2O. Draw the Lewis structure of formaldehyde.
Solution:
Step 1: Find the total number of valence electrons.
Carbon has 4 valence electrons
Hydrogen has 1 valence electrons
Oxygen has 6 valence electrons
Total valence electrons = 1 carbon (4) + 2 hydrogen (2 x 1) + 1 oxygen (6)
Total valence electrons = 12
Step 2: Find the number of electrons needed to make the atoms "happy"
Carbon needs 8 valence electrons
Hydrogen needs 2 valence electrons
Oxygen needs 8 valence electrons
Total valence electrons to be "happy" = 1 carbon (8) + 2 hydrogen (2 x 2) + 1 oxygen (8)
Total valence electrons to be "happy" = 20
Step 3: Determine the number of bonds in the molecule.
number of bonds = (Step 2 - Step 1)/2
number of bonds = (20 - 12)/2
number of bonds = 8/2
number of bonds = 4
Step 4: Choose a central atom.
Hydrogen is the least electronegative of the elements, but hydrogen is rarely the central atom in a molecule. The next lowest electronegative atom is carbon.
Step 5: Draw a skeletal structure.
Connect the other three atoms to the central carbon atoms. Since there are 4 bonds in the molecule, one of the three atoms will bond with a double bond. Oxygen is the only choice in this case, since hydrogen only has one electron to share.
Step 6: Place electrons around outside atoms.
There are 12 valence atoms total. Eight of these electrons are tied up in bonds. The remaining four complete the octet around the oxygen atom.
Each atom in the molecule has a complete outer shell full of electrons. There are no electrons left over and the structure is complete. The finished structure appears in the picture at the beginning of the example.
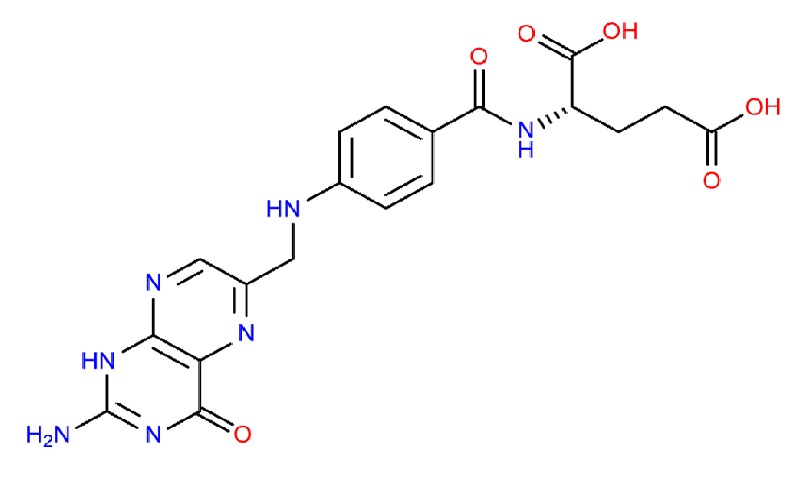
Molecular structure of the atom of some vitamins and minerals
-Yara Ceryl G. Sangco-
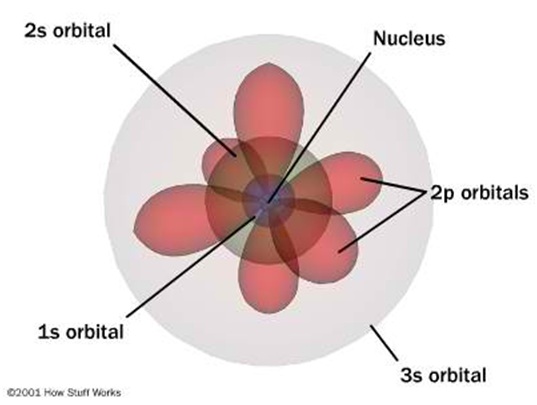
Quantum model of an atom
Bohr Model
The Bohr modeldepicts the atom as small, positively charged nucleussurrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics, and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems, the Bohr model is still commonly taught to introduce students to quantum mechanics, before moving on to the more accurate, but more complex, valence shell atom.
Quantum Mechanical Model
The quantum mechanical model is based on mathematics. Although it is more difficult to understand than the Bohr model, it can be used to explain observations made on complex atoms. A model is useful because it helps you understand what’s observed in nature. It’s not unusual to have more than one model represent and help people understand a particular topic. The quantum mechanical model is based on quantum theory, which says matter also has properties associated with waves. According to quantum theory, it’s impossible to know the exact position and momentum of an electron at the same time. This is known as the Uncertainty Principle. The quantum mechanical model of the atom uses complex shapes of orbitals (sometimes calledelectron clouds), volumes of space in which there is likely to be an electron. So, this model is based on probability rather than certainty.
The Bohr modeldepicts the atom as small, positively charged nucleussurrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics, and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems, the Bohr model is still commonly taught to introduce students to quantum mechanics, before moving on to the more accurate, but more complex, valence shell atom.
Quantum Mechanical Model
The quantum mechanical model is based on mathematics. Although it is more difficult to understand than the Bohr model, it can be used to explain observations made on complex atoms. A model is useful because it helps you understand what’s observed in nature. It’s not unusual to have more than one model represent and help people understand a particular topic. The quantum mechanical model is based on quantum theory, which says matter also has properties associated with waves. According to quantum theory, it’s impossible to know the exact position and momentum of an electron at the same time. This is known as the Uncertainty Principle. The quantum mechanical model of the atom uses complex shapes of orbitals (sometimes calledelectron clouds), volumes of space in which there is likely to be an electron. So, this model is based on probability rather than certainty.
Carmina S. Ortiz
Redox Reaction
4Ag+O2 à2Ag2O
Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid-base reactions. Fundamentally, redox reactions are a family of reactions that are concerned with the transfer of electrons between species.
OXIDATION NUMBER – the value assigned to an atom or ion in order to keep track of elecxtons gain or lost.
RULES IN ASSIGNING THE OXIDATION NUMBERS
· Elements in uncombined states have an oxidation number zero.
E.G.=Na, Mg, Al, O2
· Monoatomic ions have an oxidation number equal to their ionic charges.
E.G.=P+5=+5 Mn+2=+2 O-2=-2
S-2=-2 H+1=+1
· Hydrogen is assigned in oxidation number of +1 in most compounds and ions.Metal hydrides such as LiH and CaH2 are exceptions, the oxidation number is -1.
+1 -2
H2Oà 2+(-2)=0
· Oxygen is assigned an noxidation number of -2 in most compounds and iond. Peroxides compounds such as H2O2 are exceptions. The oxygen number of oxygen is -1.
+1 -1
H2O2à2+(-2)=0
· In molecular compounds, the more electronegative is assigned a negative oxidation state equal to its charge as an anion.
-1
CCl4à4+(-4)=0
· Ionic compounds are electrically neutral. Hence, the sum of the individual oxidation numbers is qual to the charge of the ion.
-1
BaCl2à2+(-2)=0
· Polyatomic ions are electrically charged. Therfore, the sum of the individual oxidation numbers is equal to the charge on the ion.
X -2 -2
SO4à x+(-8)=2
x= -2+8
x=+6
BALANCING REDOX REACTIONS
OXIDATION- the process in w/c a substance loses e-. It is characterized by the increased in oxidation number.
REDUCTION- the process in w/c a substance gain e-. It is characterized by decrease in oxidation number
OXIDIZING AGENT- substance that has abiliy to oxidize another substance.
REDUCING AGENT- substance that has ability to reduce another substance.
ELORA- electron loss, oxidized, reducing agent
GEROA-gain electon, reducede, oxidizing agent
Mg + 2HCl à MgCl2 + H2
0 +1 -1 +2 -1 0
Oxidation loses 2e-
Reduction gain 1e-x 2e-=2e-
0+ 2(0) = 0+0
0=0
Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid-base reactions. Fundamentally, redox reactions are a family of reactions that are concerned with the transfer of electrons between species.
OXIDATION NUMBER – the value assigned to an atom or ion in order to keep track of elecxtons gain or lost.
RULES IN ASSIGNING THE OXIDATION NUMBERS
· Elements in uncombined states have an oxidation number zero.
E.G.=Na, Mg, Al, O2
· Monoatomic ions have an oxidation number equal to their ionic charges.
E.G.=P+5=+5 Mn+2=+2 O-2=-2
S-2=-2 H+1=+1
· Hydrogen is assigned in oxidation number of +1 in most compounds and ions.Metal hydrides such as LiH and CaH2 are exceptions, the oxidation number is -1.
+1 -2
H2Oà 2+(-2)=0
· Oxygen is assigned an noxidation number of -2 in most compounds and iond. Peroxides compounds such as H2O2 are exceptions. The oxygen number of oxygen is -1.
+1 -1
H2O2à2+(-2)=0
· In molecular compounds, the more electronegative is assigned a negative oxidation state equal to its charge as an anion.
-1
CCl4à4+(-4)=0
· Ionic compounds are electrically neutral. Hence, the sum of the individual oxidation numbers is qual to the charge of the ion.
-1
BaCl2à2+(-2)=0
· Polyatomic ions are electrically charged. Therfore, the sum of the individual oxidation numbers is equal to the charge on the ion.
X -2 -2
SO4à x+(-8)=2
x= -2+8
x=+6
BALANCING REDOX REACTIONS
OXIDATION- the process in w/c a substance loses e-. It is characterized by the increased in oxidation number.
REDUCTION- the process in w/c a substance gain e-. It is characterized by decrease in oxidation number
OXIDIZING AGENT- substance that has abiliy to oxidize another substance.
REDUCING AGENT- substance that has ability to reduce another substance.
ELORA- electron loss, oxidized, reducing agent
GEROA-gain electon, reducede, oxidizing agent
Mg + 2HCl à MgCl2 + H2
0 +1 -1 +2 -1 0
Oxidation loses 2e-
Reduction gain 1e-x 2e-=2e-
0+ 2(0) = 0+0
0=0